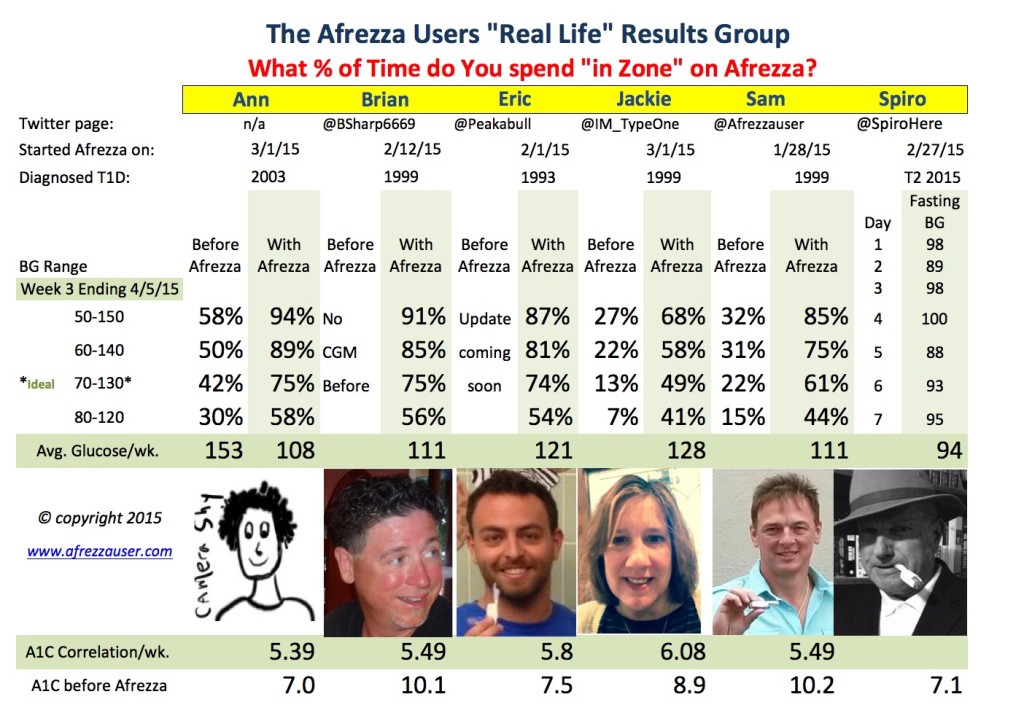
Yes, it’s Easter Week..lots of candy, snacking and family “get-togethers”–Absolutely No Problem for a diabetic taking Afrezza as you can see below! These “time in zone” measurements are life-changing for a diabetic–higher quality life=more energy, feeling better, no more chasing highs and lows all day long.
What surprises you most about the third week data?
The biggest surprise for me was how well a One Month Afrezza User “dialed it in” this week and improved on her numbers (Ann). She did an outstanding job staying in zone like never before and the correlation on her A1C this week was an astounding 5.39 (ie. non-diabetic) with an average blood glucose of 108–she beat us all! Next week I will reformat the chart and post weekly comparisons but for now it should be so obvious that Afrezza works, especially in looking at the week 3 data! Add in all the conveniences from discreet, anywhere use, pain-free inhalation vs. needles, ultra rapid effectiveness and Afrezza shows it’s superiority. Also, the A1C’s levels we are running at now are considered “non-diabetic” or “pre-diabetic”. Of course, this number needs to be run for 12 weeks before any of the participants can claim an A1C in 5’s or low 6’s. You should know that none of us have ever had an A1c that low. We are all stunned, as will our doctors be when we do get that 3 month A1C number. Note: Dr. Bode did look at my data from my Dexcom Monitor and it was only for the first 4 weeks and used the word “unbelievable” several times. On the trials I finished with an A1C of 7.1 even with all the protocols. Can’t wait to see other doctors reaction around the country when all the Afrezza users officially report their #’s – doctors will be shocked.
What is the Afrezza Users “Real Life” Results Group?
It is a group of Afrezza Users who use a Continuous Glucose Monitors (Type 1’s) seeking tighter diabetes control and measuring the time spent in each blood sugar range. It is NOT a competition and was only started so people could understand and see the inherent benefit of why Afrezza provides better control and higher patient satisfaction for t1 and t2 diabetics over our previous regimens.
Why are you doing this?
There has been so much discussion about the FDA Afrezza Drug trials and why I feel as a participant in the Affinity 1 trial, there were significant “hold backs” as to why we were not able to materially outperform Injectable Insulin because of the protocols on the trials. This is no longer the case!
To Summarize just a few:
- Afrezza Dosing only changed approx. every 6 weeks, not flexible depending on meal
- No CGM’s allowed
- Time in Zone not measured
- Patient quality of life not measured
- Brand new users vs. Very experienced users (what if it was brand new injectable users vs. experienced Afrezza users—would that be fair? No-they would stop the trial right away because it would be a TKO/referee stops contest victory for Afrezza which we will prove by real world experience.)
What would you like to express to everyone out there considering Afrezza or to any doctors considering prescribing Afrezza for their patients?
My opinion is that Afrezza is a “life-changing” drug that is not very well understood by the diabetic community at large—both doctors and patients. It improved my life and gave me hope in treating my diabetes. I would wish the same for any other diabetic out there—a better and higher quality of life!

Thanks Sam for your diligent work in updating the stats. Quite an impressive pattern is forming showing the unique benefits of Afrezza over a longer period of time.
I think you are all fabulous people with no financial interest in the company and only want to encourage other diabetics to give this a try If it works for them fine
If it doesn’t no one is holding back anyone from returning to their previous treatment. Al Mann said the true test was going to take place when it it becomes available to the public.It is now available. I applaud you all for your courage and perseverance.
i wish you all healthy and long lives.
Hi Sam! I’ve been following your posts with much enthusiasm. Please continue to compile more information as you are able to. My uncle overseas is a T2 who I feel could benefit greatly from Afrezza, but am hesitant about recommending anything to him prematurely. Thanks to you and the other early adopters for your hard work and reporting!
What long-active insulin are you guys using? I am on Lantus is now. Do you recommend something different?
I have my next appointment with my doctor in a few days and will be discussing this with him. I would say if something is working great for you perhaps stick with it. Of course you should check with your doctor to see if there are any reasons to switch to a new product. I can tell you that Lantus worked best on a split dose.