Week 11 already and June is here. Around the corner is the ADA Meeting in Boston beginning on Friday June 5th at 11:45am. The following Afrezza presentation will take place on Sunday, June 7th from 10:15am-11:00am:
Location: Product Theater 2 (Booth 865).
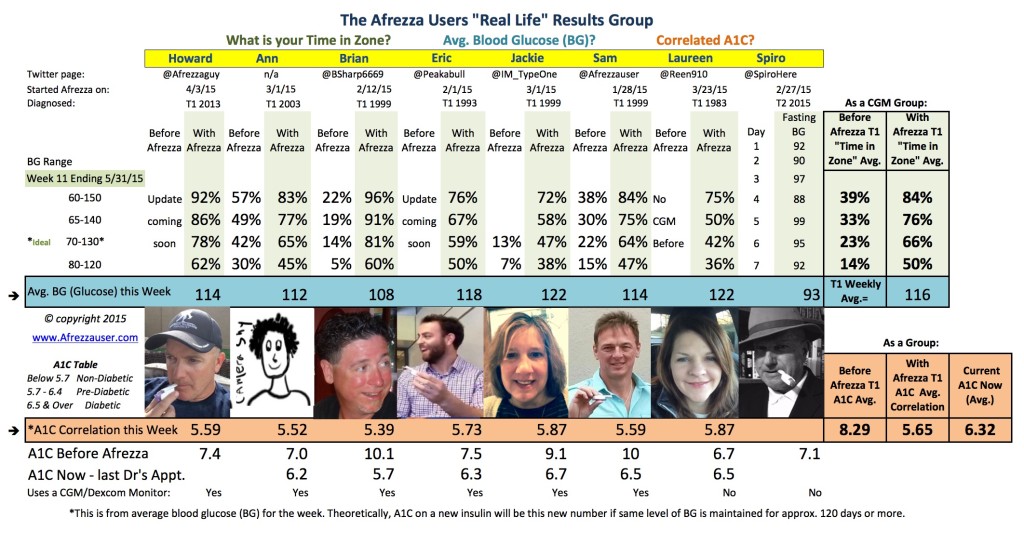
Within our group everyone is measuring A1C Correlations in the 5’s this week–yes, it’s really no surprise any longer – tighter control and ongoing improvement have become the new normal!
This Afrezza journey has been remarkable. And every single one of us has completely had a turnaround for what is acceptable as a diabetic–and our “Time in Zone” and “Blood Glucose” numbers clearly reflect that. For most of us, our upper limits on our Dexcom CGM’s went from 220 or higher down to 120, which is truly remarkable.
Here are the numbers for this week:
What surprises you most about the Week 11 data?
To tell you the truth, I am not surprised at our ongoing data. We’re almost 3 months in, and we’ve all got Afrezza “dialed in”, which in contrast to Novolog, which many of us used to take, was never “dialed in”, even after 10 to 20 years of trying! Perhaps the most surprising thing is that more diabetics haven’t run to their doctors to request Afrezza. It is clearly only a question of time given the amazing results we are all experiencing.
What is the Afrezza Users “Real Life” Results Group?
It is a group of Afrezza Users who use a Continuous Glucose Monitors (Type 1’s) seeking tighter diabetes control and measuring the time spent in each blood sugar range. Laureen uses “finger prick” measurements throughout her day to measure her blood glucose levels while the rest of us are using CGM’s (continuous glucose monitors). It is NOT a competition between T1’s and T2’s nor among any of us and was only started so people could understand and see the inherent benefit of why Afrezza provides better control and higher patient satisfaction for t1 and t2 diabetics over any our previous regimens. In essence, this is a “real-world” trial of “do your best”, and have good and bad days, but keep track of it all, and let everyone know the results. Well, the results thus far are clearly unbelievably positive.
Why are you doing this?
There has been so much discussion about the FDA Afrezza Drug trials and why I feel as a participant in the Affinity 1 trial, there were significant “hold backs” as to why we were not able to materially outperform Injectable Insulin because of the protocols on the trials. This is no longer the case!
To Summarize here are just a few of the hold backs why Afrezza didn’t get to show how great it works:
- Afrezza Dosing was only changed approx. every 6 weeks, and not flexible dosing depending on meal
- No CGM’s allowed
- Time in Zone not measured
- Patient quality of life not measured
- Brand new Afrezza users vs. Very experienced Injectable users (what if it was brand new injectable users vs. experienced Afrezza users—would that be fair? No-they would stop the trial right away because it would be a TKO/referee stops contest victory for Afrezza which we are already proving by “real-world” experience.)
What would you like to express to everyone out there considering Afrezza or to any doctors considering prescribing Afrezza for their patients?
My opinion is that Afrezza is a “life-changing” drug. It improved my life and gave me hope in treating my diabetes. I wish the same for you or any other diabetic you know—enjoy a better and higher quality of life! Please demand to try Afrezza from your doctor, and if he/she won’t or is not able to prescribe it–go find one that will–it’s your life!

Congrats and thank you all for the insight and info!
Congrats to all volunteers for your ongoing commitment in sharing your readings on Afrezza. You are all doing some service to others with diabetes, by allowing them to see in ‘real time’ how Afrezza, the new inhaled insulin continues to display superior remarkable positive effects in controlling meal-time blood glucose spikes. Thanks to you all, and a big thank you to Sam Finta aka afrezzauser.